Douglas G. Pfeiffer, Dept. Entomology, Virginia Tech, Blacksburg VA 24060, dgpfeiff@vt.edu
There are two main spider mite pests that affect fruit crops in the east, but the most important one in our small fruit crops is the twospotted spider mite (TSM), Tetranychus urticae. This species is whitish to straw colored, with two large black spots. These spots are actually the gut contents seen through the cuticle. Each time the mite molts during its development, the gut contents are voided, so there are some brief intervals when the mites are lacking their spots! The eggs are spherical, and clear translucent (Fig. 1).
The host range of twospotted spider mite is very broad – feeding on many distantly related plant species. This makes this polyphagous mite the most agriculturally important mite in the world. TSM overwinters as adults in the ground cover, showing up in the canopy later in the season. The total life cycle requires only 8-12 days. The average fecundity, or eggs per female, also goes up – usually 90-100 eggs, but up to 200. And in the fall, you may notice another difference.
There is an overwintering morph that looks quite different. The adult mites become bright orange, with no spots. Figure 2 shows a group of mites that have gathered at the calyx of an apple just before harvest.
In humid parts of the country, like our southeastern states, the usual spider mite pest in orchards and vineyards is European red mite (ERM), Panonychus ulmi. This mite appears a dark, brick-red color with white spots or tubercles. A related pest of citrus, citrus red mite, looks similar but without the white tubercles. Even on these fruit crops, TSM will often predominate in hot, drought years.
ERM overwinters on tree bark in the egg stage. In cases of severe infestations, those reddish eggs can give the undersides of scaffold limbs a reddish color. These overwintering eggs hatch from the pre-pink through bloom of apple. The mites then move to leaves to begin their development. The first stage out of the egg is the larva – a 6-legged stage, not much bigger than the egg itself. It only lasts a day or so. This is followed by two nymphal stages, both 8-legged. These are the protonymph and deutonymph stages. Finally, the 8-legged adults are produced. The adult female is oval and dome-shaped. The male tapers to a blunt point at the hind end.
The life cycle of ERM takes 20-25 days – less time, as little as 10-12 days when it’s hot. Females live about 18 days, and lay about 35 eggs each. I’ll come back to these figures in a little while. There can be 10-12 generations annually. The overwintering eggs start to appear fairly early, in mid-August.
Let’s take a few seconds to summarize the differences between TSM, the usual spider mite in small fruit crops, and ERM, the usual spider mite pest in vineyards and orchards. TSM develops faster than ERM – 10 days versus 25 days. TSM has a higher reproductive rate – 100 eggs per female compared with 35. In addition, TSM has a more severe impact on leaf function, making it more injurious on a per-mite basis. This is probably the result of having to feed more to fuel the faster development and greater reproduction. Consequently, we should cut in half any action thresholds that were developed for the more usual ERM. Another thing to keep in mind is that TSM can be harder to kill. If TSM is the predominant spider mite, and there is a rate range on an acaricide label, use the upper end of the rate range.
The photo in Fig. 3 shows a male ERM waiting for the adult female to emerge from the deutonymph stage. The same behavior is seen in TSM. At this time, the female produces a sex pheromone that attracts the male to her side. As soon as she emerges, mating will take place. Spider mites have an unusual type of reproduction, that is probably one of the several factors that contribute to this family being the leading arthropod group for pesticide resistance. They show a mode of reproduction called arrhenotoky, or haplodiploidy. This means that if females are mated, and the eggs fertilized, they give rise to female offspring. These are diploid, with genes contributed by both parents. If a female remains unmated, and the eggs don’t get fertilized, she gives rise to male offspring. These are haploid, with genes from the mother alone. So if a lone female survives an acaricide spray because she happens to be resistant, she doesn’t have to find a mate – she can produce offspring anyway, and being haploid, these WILL carry her genes for resistance. And yet if mating is available, that will allow for the recombining of genes that is great for the formation of new strains. These mites are adapted for both rapid population growth, AND recombining genes.
Spider mites are usually secondary pests. They rise to pest status when natural enemies are removed from the system, usually by sprays. Now let’s look at some of the predators that feed on spider mites. There is a complex of predatory mites, mostly in the family Phytoseiidae. In commercial orchards, the most common one in our area is Neoseiulus fallacis. You may see it called Amblysius fallacis, an older name. These mites are teardrop-shaped, and flatter in appearance than spider mites. They are also much more active, as you would expect from a predator.
Neoseiulus females lay about 100 eggs each. Development of immatures takes about 8 days, and they live about a month as adults. Their favored prey is spider mites but they can sustain themselves on other mites or pollen. They do best at 50-80 degrees F, but can tolerate higher temperatures if there is high humidity. They do well feeding on mites on strawberry. Remember that they overwinter in the ground cover, so they are already in close proximity to the crop!
Neoseiulus follows two different hunting strategies, depending on prey density. At high prey density, they follow a random path. This is likely to bring them in contact with prey. At low prey density, they follow leaf edges, and they may attempt to disperse aerially. If starving, they will face into the breeze, lift their front end, and allow themselves to be carried off. Growers can estimate predator prey rations to determine likelihood of successful biological control. If there is a ratio of 1:10 – 1 fallacis for every 10 ERM, there is a good likelihood of control. Higher ratios increase the chances of successful control. Even 1:20 can be successful on some varieties.
There is an important way of categorizing biological control. At the upper level, we can distinguish between conservation biological control, and augmentation of natural enemies. In conservation biological control, we aim to protect naturally occurring populations of predators and other natural enemies. This is mainly done through selection of pesticides that are least toxic to the beneficial species. Under augmentation, natural enemies may be increased, either by an inoculative release (releasing a few with the goal of establishment) or by an inundative release (releasing large numbers of natural enemies to overwhelm the pest population, whether or not the predators successfully establish).
Neoseiulus is available commercially from several insectaries. It is very helpful to explore the web sites and online catalogs of these insectaries – they contain biological information that is helpful in selecting the right predator for your situation, and other ways of making your plan successful. Here is a non-exclusive list:
- Green Methods – https://greenmethods.com/
- IPM Labs – https://www.ipmlabs.com/
- Koppert – https://www.koppertus.com/
- ARBICO – https://www.arbico-organics.com/
Let’s say a few words about chemical control of mites. In the 1980s and into the 90s, we were in a tough spot – effective chemical had dwindled to a very few. In fact, in some orchards, because of resistance history, there no compounds that were very effective. Now we are in a much better position. We have acaricides in several mode of action classes, shown by the IRAC number on the label (Insecticide Resistance Action Committee). Since spider mites are so prone to develop resistance, it is important to rotate among different MOA classes. This is an important part of a resistance management plan. It is also important to select materials of the lowest toxicity to predators possible – this applies to other insecticides directed against other pests, as well as acaricides. By making life easier for natural enemies, we can also lower the need for chemical intervention, shepherding the useful life of effective materials.
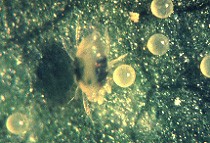
Twospotted spider mite female and eggs. (Photo Mid-Atlantic Orchard Monitoring Guide)
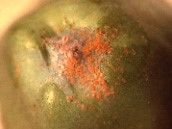
Overwintering morph of twospotted spider mite.

Adult male European red mite waiting for female to emerge after molting.
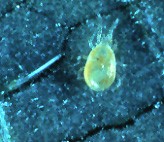
Neoseiulus fallacis, a predator of spider mites. (Photo Mid-Atlantic Orchard Monitoring Guide)