Alejandro Rojas, Dept of Plant Pathology, University of Arkansas
Strawberry is one the most widely cultivated fruits in the US and it is a major berry crop in Arkansas, with at least 30 growers around the state, or in the nearby states. Soil infestation by plant pathogens in strawberry fields are amongst the most limiting diseases in this production system. The methyl bromide transition program has reduced the number of tools to manage soil borne diseases. Other practices such as plasticulture have been used to promote other aspects of the crop such as yield and weed management; however, these practices could not have an effect on soilborne pathogens. In Arkansas, the production system is based on annual plants aiming to rotate crops to avoid pathogen build -up and manage other pests. However, growers have limited areas for production that reduces the options for rotation. Having this in mind, it was important to characterize what potential fungal and oomycetes (water molds) pathogens were prevalent in growers’ fields in the region. The identification of potential problems in soil could help to establish strategies to reduce impact of those soilborne pathogens.
One of the problems that growers faced with strawberries and soilborne diseases is the difficulty to address the symptoms and relate those to a specific pathogen, since most of the symptoms are often wilt, root rot, crown rot and plant death. Among the pathogens causing these symptoms, there are four common pathogens causing damage on strawberries. Phytophthora cactorum and P. fragariae (water molds) causing crown rot and root rot; Rhizoctonia solani causing black root rot; Macrophomina phaseolina causing a charcoal rot, and Fusarium oxysporum causing wilting (Maas 1998). To establish the prevalence of soilborne pathogens in Arkansas and nearby regions, plant and soil samples from strawberry fields were submitted by growers or collected and processed for the isolation of pathogens.
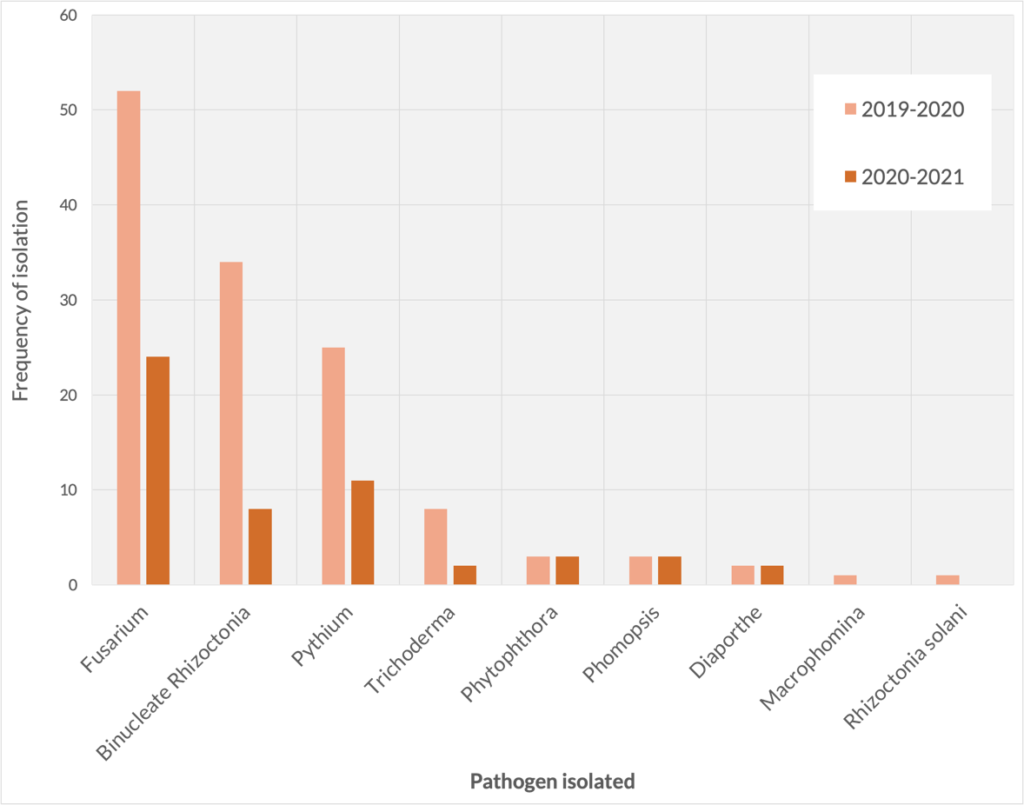
Throughout 2019-2020, composite soil samples and plants were taken from 20 strawberry farms around Arkansas and neighboring states. From plant tissue, isolations were done on Potato Dextrose Agar (PDA) and Corn Meal Agar (CMA-PARP) and soil samples were processed for DNA extraction were analyzed for quantity and variety of fungal and oomycete pathogens. In 2019-2020, eighteen submissions were processed and in 2021-22, ten samples were processed (Figure 1). If isolated from plant tissue, crown, runners and roots were assessed. For soil samples, we relied on isolation by baiting (Spurlock et al. 2015). From the results, Fusarium is widely prevalent in samples in both years, followed by binucleate Rhizoctonia and Pythium. Most of these pathogens are widely present, especially Fusarium, which is a common soil inhabitant, however, it is important to clarify that not all Fusarium could be pathogenic. While Rhizoctonia and Pythium are more likely to cause disease in seedlings and growing plants. These pathogens are typically called “root nibblers” (Figure 2), and their prevalence and incidence get worse due to multiple years of production in the same bed. It is also common to have these pathogens causing disease problems as complex.

Diagnostics from these samples often requires isolation like it was done for the survey, and diagnosis based of symptoms could be difficult. Samples could be submitted to the Arkansas Plant Health Clinic to get a more accurate diagnosis. We also implemented molecular diagnostics, similar to the ones used during the pandemic, however we are aiming to detect Phytophthora from the soil samples and also have an idea of how prevalent it could be on soils in Arkansas (Miles et al. 2017). Phytophthora could be a damaging pathogen causing root rot but also affecting fruit quality. From isolations, we were able to recover less than five isolates, but using the molecular diagnostics in soil, all soil samples were below the detection threshold, which means that at least in 2020 and 2021, Phytophthora was not prevalent in the fields, however that could change over time.
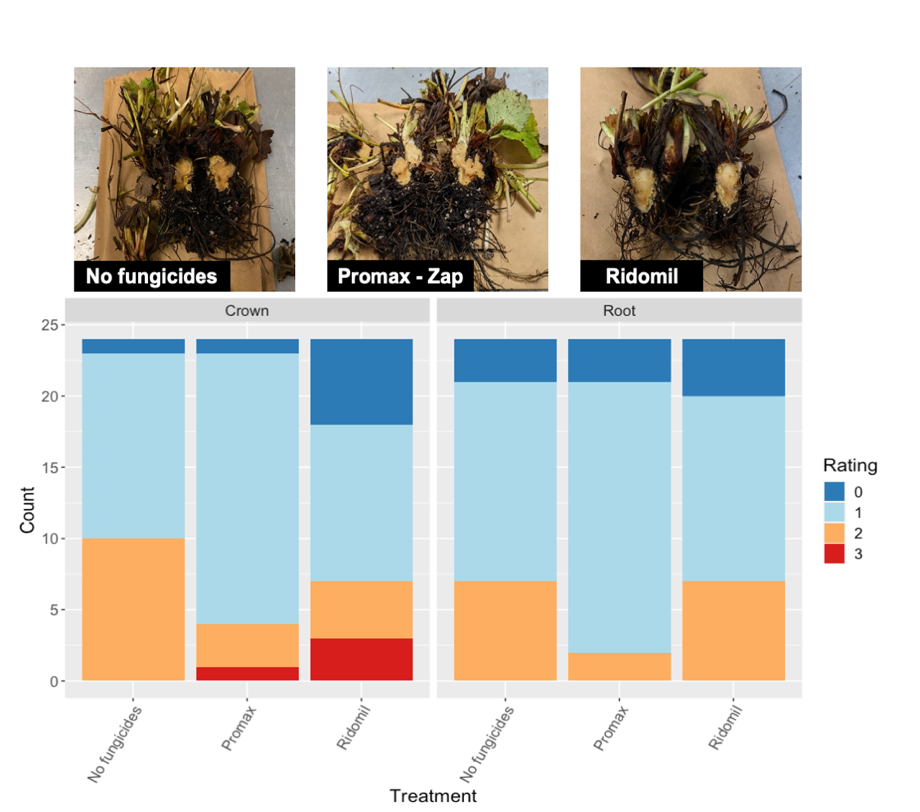
In addition, a research plot was established in at Vegetable station (Kibler, Arkansas) in collaboration with Dr. McWhirt and Dr. Cato. Three plots with strawberry cv. Chandler were planted in September 2020 and soil samples were collected at different stages to determine levels of soil microbial communities and to evaluate impact of management practices used by growers in the region. Plots received three different management strategies using chemigation: Promax+ Zap, Ridomil, and untreated (No fungicide). In terms of disease, plants were scored for crown and root rot using severity scale 0-3 (0 = healthy root/crown tissue and 4 = severe root and crown rot). Overall, there are not strong differences across treatments, however, Ridomil Gold (ai. mefenoxam) had a greater number of plants with healthy roots and crowns. The trial is being conducted again this season and more information will be available about prevalence of soilborne pathogens under the different management strategies. Isolation from the plant samples had the usual suspects: Fusarium, Macrophomina, Pythium and Rhizoctonia. All pathogens were consistent across treatments.
One of the issues in Arkansas production system is the use of transplants, and plugs could be an entry point or carrier for pathogens that are not present in a field. Therefore, pre-planting management is important to reduce chances of disease, by using pre-plant dips or selecting tolerant varieties. Other strategies to reduce impact of diseases is the selection of fields or using beds to avoid low lying and poorly drained soils, as well as the crop rotation to avoid build up. The use of organic matter to improve soil texture is another strategy, but the most effective way to improve the overall soil health is the use of cover crops that will improve microbial activity and increase organic matter. It is also important to consider the application and timing of fungicide applications, since those could help to control pathogens, but they could also affect beneficial microbes, reducing chances of nontarget effects.
References
Maas, J. L. 1998. Compendium of Strawberry Diseases, Second Edition. The American Phytopathological Society. Available at: https://apsjournals.apsnet.org/doi/10.1094/9780890546178.
Miles, T. D., Martin, F. N., P, R. G., Bilodeau, G. J., and Coffey, M. D. 2017. Systematic Development of Phytophthora Species-Specific Mitochondrial Diagnostic Markers for Economically Important Members of the Genus. Plant Disease. 101:1162–1170.
Spurlock, T. N., Rothrock, C. S., and Monfort, W. S. 2015. Evaluation of Methods to Quantify Populations of Rhizoctonia in Soil. Plant Disease. 99:836–841.