By Douglas G. Pfeiffer, Dept. Entomology, Virginia Tech, Blacksburg VA 24060, dgpfeiff@vt.edu
Spotted-wing drosophila (SWD), Drosophila suzukii (Matsumura), was first introduced into the US in 2008, first reaching the western states from Asia. It reached the Southeast in 2009, and continued to spread through much of the US through 2012, SWD has presented a huge problem for berry growers. This pest has been discussed recently in Small Fruit New, regarding both conventional and organic management (Pfeiffer 2020, Sial 2020).
SWD is a difficult pest to control. Insecticide resistance is a likely obstacle because of the number of eggs produced by females, and the number of annual generations, but factors that are conducive to resistance. Growers should use a combination of tactics to take the pressure off chemical control.


Hosts
All berry crops are suitable hosts for SWD, with caneberries and blueberries most vulnerable. SWD does not survive as well in grape; nevertheless, females may lay eggs in grape, with subsequent development of sour rot.
Description
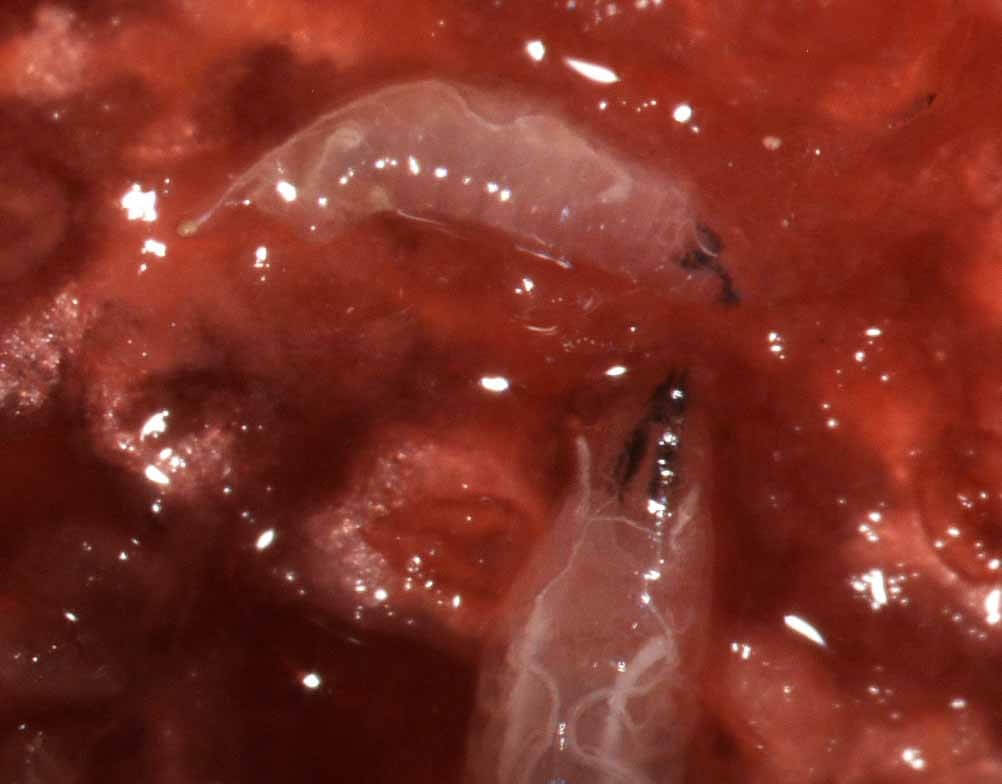
By now, most berry growers are familiar with this insect and itenabless appearance. Male SWD is differentiated from native Drosophila species by the presence of a black spot on the leading edge of the wing (Fig. 1a); females lack this spot but have a large serrated ovipositor, visible on the underside of the abdomen (Fig. 1b). This structure allows the insect to insert eggs into the flesh of a ripening fruit, allowing larval establishment ahead of other drosophilids, which colonize rotting or overripe fruit material. Eggs are white, elongate oval, inserted under the skin of the berry, with long respiratory filaments from one end (Fig. 2a). These respiratory horns may be found protruding from an oviposition site with magnification (Fig. 2b).
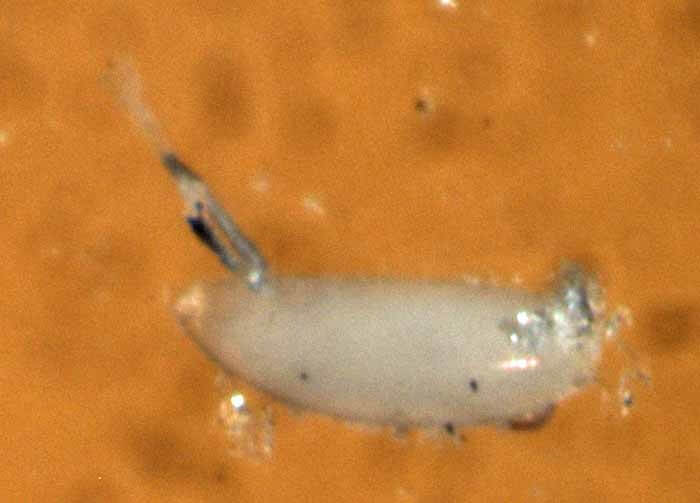

Larvae are translucent maggots 2-3 mm long, with black mouth hooks visible at the anterior end (Fig. 3a). Silvery white tracheal tubes may be visible through the dorsal cuticle with magnification. Respiratory projections are present on the posterior end, giving an appearance of being pointed at each end. Puparia (covering of the actual pupa) (Fig. 3b) are brown, elliptical, about 3 mm long, with respiratory projections from the hind end.

Biology
In eastern Asia, there are up to 13 generations. A life cycle can be completed in 8-14 days, but adults can live up to 9 weeks. Females use the atypically large ovipositor to lay eggs in fruits as they are ripening, earlier than other drosophila species. Eggs are inserted under the skin of ripening fruit; each female lays 7-16 eggs/day. Eggs hatch in 1-3 days, and larval feeding on the flesh causes a collapse of localized tissue after another 2 days, followed by growth of fungal or bacterial organisms.
Monitoring
Traps should be used to detect activity, and when flies are detected, make sure that other control measures are in place. Traps are not effective in providing control. Several commercial traps are available (Trece and Scentry). A trapping guide has been posted (Wallingford et al. 2018), with discussion of several baits, and comparing commercial with homemade traps.
Control
Chemical Control
Control measures are directed against the adults; there are no effective controls for larvae in the fruit. As vulnerable fruit approach ripeness, weekly spray applications should be made. Because of the high number of offspring and number of generations, there is a high risk of development of insecticide resistance. Consequently, insecticides with different mode of actions should be rotated to prolong the effective life of insecticides. For more details on conventional and organic chemical control, click on the link for Small Fruit News Summer 2020 edition.
Our Southern Region Small Fruit Consortium provides recommendations for SWD in the pest management guides for caneberries, blueberries, strawberries and bunch grapes. In addition, individual states may maintain small fruit management guides that are helpful.
- Caneberries: https://smallfruits.org/files/2020/12/2021-Caneberry-Spray-Guide.pdf
- Blueberries: https://smallfruits.org/files/2021/01/2021-Blueberry-Spray-Guide.pdf
- Strawberries: https://smallfruits.org/files/2020/12/2021-Strawberry-IPM-Guide.pdf
- Bunch grapes: https://smallfruits.org/files/2021/02/2021-Bunch-Grape-Spray-Guide.pdf
Cultural Control
Netting of 80g weight was effective in controlling injury by SWD (McDermott and Nickerson 2014, Leach et al. 2016, Riggs et al. 2016, Ebbenga et al. 2019). Lighter grades (larger mesh) are not effective. While netting is initially expensive, it becomes cost effective because it may be used for several years.
Harvest fruit promptly and thoroughly to eliminate breeding sites. It is important to harvest all fruit, including those in the interior and lower parts of the plant canopy. This can be problematic in pick-your-own operations. This issue should be kept in mind once SWD established in an area, since at times grape growers may leave berries on the vine to allow greater development of some harvest parameters. Any overripe or rotten fruit nearby should be destroyed. In vineyards, pomace produced during the crushing process should not be dumped near the producing vineyard block. This can become a source for many SWD.
When berries are harvested, it will be helpful to get them as cool as possible, as soon as possible. There is complete mortality of larvae in fruit held for 96 hours at 35˚F, and below 40˚F, eggs and larvae don’t develop (Bolda 2010, Burrack 2016). In most cases, such uniform holding conditions are not maintained; fruit cooling should be considered a component of SWD management and not a sole control tactic.
Biological Control
Because of the ability of SWD to encapsulate and kill the eggs of our native parasitoid wasps, biological control has not been successful. Research is underway to find parasitic species that are able to attack this species.
Recent Research
Because the importance of SWD to berry producers continues, there is a lot of interesting research going on, both in the US and internationally. It beyond the scope of this newsletter article to provide a complete review, but here are some examples. The geographic spread of SWD continues, and there has been a recent first report of injury to berries in Africa, in northwestern Morocco (Boughdad et al. 2021). Researchers continue to delve into the basic biology of SWD. A recent review of chemical ecology (Cloonan et al. 2018) pointed out that current food-odor traps are unlikely to be sufficiently attractive to SWD, and further work on compounds that are attractive and aversive is needed. Nutritional ecology research continues. In a behavioral study (Young et al. 2018), the role of protein and carbohydrate content in female feeding and ovipositional choices was examined. This could lead to improved monitoring strategies. Guedes et al. (2019) used electropenetrography to compare probing, feeding and egg laying behavior in media and strawberry. Landscape level studies continue to inform our SWD ecological understanding. Dropped and waste fruit proved to be an important source of off-season development of SWD, with higher numbers of SWD near cideries and wineries (Bal et al. 2017). A study on spatial ecology in the landscape (Santoiemma et al. 2019) indicated that the risk of SWD outbreaks depends on factors beyond the control of traditionally scaled management. Competition, both intraspecific (Bezerra Da Silva et al. 2019a) and interspecific (Shrader et al. 2020) can be important to SWD. Exposure in intraspecific competition influences pupation site in SWD, with larvae exposed to competition traveling farther to pupate. Interspecific competition with another exotic drosophilid, African fig fly, Zaprionus indianus, can affect SWD mortality and developmental time.
Of course, applied research on SWD management continues as well. Biocontrol has been problematic, as indicated above. Recent work has indicated that the pupal parasitoid Pachycrepoideus vindemmiae still presents hope in improving biological control of SWD (Bezerra Da Silva et al. 2019b). Lee et al. (2019) provided a review of biocontrol work, and will be a useful source of information wishing to catch up here. This review included both parasitoids and fungal and bacterial pathogens. There is ongoing research on cultural control as well. For example, Leach et al. (2018) examined the effect of shortening harvest intervals in raspberries. A two-day harvest schedule result3ed in lower SWD infestation than 3-day harvest interval. Sterile Insect Technique (SIT), a technique that has been successful in a range of other pests, is explored for SWD (Sassù et al. 2019). SIT has also been combined with the use of incompatible Wolbachia symbiotes (Nikolouli et al. 2020). When males and females are infected with different strains of Wolbachia, there has been reduced mating success in other insects, including a fruit pest, plum curculio (Zhang and Pfeiffer 2008, Zhang et al. 2010).
In summary, in addition to proper pest management practices in use today, including appropriate resistance management approaches, work continues on approaches that will aid SWD management in our region.
References:
Bal, H. K., C. A. Adams, and M. Grieshop. 2017. Evaluation of off-season potential breeding sources for spotted wing drosophila (Drosophila suzukii Matsumura) in Michigan J. Econ. Entomol. 110: 2466–2470.
Bezerra Da Silva, C. S., K. R. Park, R. A. Blood, and V. M. Walton. 2019a. Intraspecific competition affects the pupation behavior of spotted-wing drosophila (Drosophila suzukii). Scientific Reports 9.
Bezerra Da Silva, C. S., B. E. Price, A. Soohoo-Hui, and V. M. Walton. 2019b. Factors affecting the biology of Pachycrepoideus vindemmiae (Hymenoptera: Pteromalidae), a parasitoid of spotted-wing drosophila (Drosophila suzukii). PLoS One 14: e0218301.
Bolda, M. 2010. Length and magnitude of fruit cooling and spotted wing drosophila mortality, Strawberries and Caneberries. Univ. Calif. Agric. Nat. Res. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=2474.
Boughdad, A., K. Haddi, N. El Bouazzati, A. Tahiri, C. El Anbri, T. Eddaya, Z. A., and A. Biondi. 2021. First record of the invasive spotted wing Drosophila infesting berry crops in Africa. J. Pest Sci. (doi:10.1007/s10340-017-0838-z) 94: 261–271.
Burrack, H. 2016. Preventing and managing spotted wing drosophila infestation. NC State Extension. Entomology – Insect Biology and Management. https://entomology.ces.ncsu.edu/2016/06/preventing-and-managing-spotted-wing-drosophila-infestation/.
Cloonan, K. R., J. Abraham, S. Angeli, Z. Syed, and C. Rodriguez-Saona. 2018. Advances in the chemical ecology of the spotted wing drosophila (Drosophila suzukii) and its applications. J. Chem. Ecol. 44: 922–939.
Ebbenga, D. N., E. C. Burkness, and W. D. Hutchison. 2019. Evaluation of exclusion netting for spotted-wing drosophila (Diptera: Drosophilidae) management in Minnesota wine grapes. J. Econ. Entomol. 112: 2287–2294.
Guedes, R. N. C., F. A. Cervantes, E. A. Backus, and S. S. Walse. 2019. Substrate-mediated feeding and egg-laying by spotted wing drosophila: waveform recognition and quantification via electropenetrography. J. Pest Sci. 92: 495–507.
Leach, H., S. Van Timmeren, and R. Isaacs. 2016. Exclusion netting delays and reduces Drosophila suzukii (Diptera: Drosophilidae) infestation in raspberries. J. Econ. Entomol. 109: 2151-2158.
Leach, H., J. Moses, E. Hanson, P. Fanning, and R. Isaacs. 2018. Rapid harvest schedules and fruit removal as non-chemical approaches for managing spotted wing Drosophila. J. Pest Sci. 91: 219–226.
Lee, J. C., X. Wang, K. M. Daane, K. A. Hoelmer, R. Isaacs, A. A. Sial, and V. M. Walton. 2019. Biological control of spotted-wing drosophila (Diptera: Drosophilidae)—Current and pending tactics. J. Integr. Pest Manag. 10: 1-10.
McDermott, L., and L. Nickerson. 2014. Evaluation of insect exclusion and mass trapping as cultural controls of spotted wing drosophila in organic blueberry production. N. Y. Fruit Quarterly 11: 25-27.
Nikolouli, K., F. Sassù, L. Mouton, C. Stauffer, and K. Bourtzis. 2020. Combining sterile and incompatible insect techniques for the population suppression of Drosophila suzukii. J. Pest Sci. 93: 647–661.
Pfeiffer, D. G. 2020. Conventional management of spotted-wing drosophila, Drosophila suzukii (Matsumura). Small Fruit News 20: https://smallfruits.org/2020/2007/conventional-management-of-spotted-wing-drosophila-drosophila-suzukii-matsumura/?cat=2025.
Riggs, D. I., G. Loeb, S. Hesler, and L. McDermott. 2016. Using insect netting on existing bird netting support systems to exclude spotted wing drosophila (SWD) from a small scale commercial highbush blueberry planting. N.Y. Fruit Quarterly 24: 9-14.
Santoiemma, G., D. Fioretto, D. Corcos, N. Mori, and L. Marini. 2019. Spatial synchrony in Drosophila suzukii population dynamics along elevational gradients. Ecol. Entomol. 44: 182-189.
Sassù, F., K. Nikolouli, R. Pereira, M. J. B. Vreysen, C. Stauffer, and C. Cáceres. 2019. Irradiation dose response under hypoxia for the application of the sterile insect technique in Drosophila suzukii. PLoS One 14: e0226582.
Shrader, M. E., H. J. Burrack, and D. G. Pfeiffer. 2020. Effects of interspecific larval competition on developmental parameters in nutrient sources between Drosophila suzukii (Diptera: Drosophilidae) and Zaprionus indianus. J. Econ. Entomol. 13: 230-238.
Sial, A. 2020. Organic management of spotted-wing drosophila (SWD) in small fruit production systems. Small Fruit News 20: https://smallfruits.org/2020/2007/organic-management-of-spotted-wing-drosophila-swd-in-small-fruit-production-systems/?cat=2025.
Wallingford, A., B. Sideman, and G. Hamilton. 2018. Monitoring spotted wing drosophila (SWD) with traps. Univ. New Hampshire Extension. https://extension.unh.edu/resource/monitoring-spotted-wing-drosophila-swd-traps.
Young, Y., N. Buckiewicz, and T. A. F. Long. 2018. Nutritional geometry and fitness consequences in Drosophila suzukii, the Spotted‐Wing Drosophila. Ecology and Evolution 8: 2842-2851.
Zhang, X., and D. G. Pfeiffer. 2008. Evaluation of reproductive compatibility of interstrain matings among plum curculio populations in the eastern United States. Environ. Entomol. 37: 1208-1213.
Zhang, X., S. Luckhart, Z. Tu, and D. G. Pfeiffer. 2010. Analysis of Wolbachia strains associated with Conotrachelus nenuphar (Coleoptera: Curculionidae) in the eastern United States. Environ. Entomol. 39: 396-405.